
Luminescence
There are many different kinds of luminescence. The mechanism is different in
each case, but some materials fit into more than one category. Some luminescence
categories are are listed in the following table:
Types of Luminescence.
| Name
| Excitation by
|
| Photoluminescence
| Photons
|
| Cathodoluminescence
| Electrons
|
| Electroluminescence
| Electric fields
|
| Triboluminescence
| Mechanical stress
|
| Chemiluminescence
| Chemical reactions
|
| Sonoluminescence
| Intense ultrasound
|
Possibly the first luminescent material ever synthesized was barium sulfide (BaS).
It was discovered by an alchemist called Vincenzo Cascariolo in 1603. Much later, calcium
sulfide was found to give much brighter luminescence. It was first created by mixing
ground up oyster shells with sulfur and heating the mixture (2CaCO3 + 3S -> 2CaS + 2CO2 + SO2).
Compounds such as CaS rely upon the inclusion of small concentrations of impurity atoms
for luminescence. In fact, if the CaS was completely pure, it would not exhibit luminescence.
These impurity atoms are incorporated into the crystal lattice of the host material and are
called a dopant. The emission characteristics are more characteristic of the dopant atoms
than of the host lattice.
 To make CaS, I mixed equal quantities of calcium carbonate and sulfur powder in a
test tube. I also mixed in a very tiny amount (less than 1%) of 'Lo Salt' to act as the
dopant. Lo Salt is manufactured by Klinge and is 66% potassium chloride and 33% sodium
chloride.
To make CaS, I mixed equal quantities of calcium carbonate and sulfur powder in a
test tube. I also mixed in a very tiny amount (less than 1%) of 'Lo Salt' to act as the
dopant. Lo Salt is manufactured by Klinge and is 66% potassium chloride and 33% sodium
chloride.
At this point the mixture, which is in the bottom of the right hand test tube, is not
luminescent.
 I heated this mixture gently over a gas burner until vapor stopped coming off and I was
left with a grey lump in the bottom of the test tube. This lump shows weak luminescence
under UV. I then transferred it to a ceramic dish, ground and mixed it again and then
heated it until it glowed orange. When cool, the compound glows with a mixture of pale
pink and green patches under UV and has an after-glow.
I heated this mixture gently over a gas burner until vapor stopped coming off and I was
left with a grey lump in the bottom of the test tube. This lump shows weak luminescence
under UV. I then transferred it to a ceramic dish, ground and mixed it again and then
heated it until it glowed orange. When cool, the compound glows with a mixture of pale
pink and green patches under UV and has an after-glow.
If you decide to try this, don't use large quantities of the chemicals. A by-product
of the reaction is sulfur dioxide gas, which is toxic.
 I also tried using strontium carbonate instead of calcium carbonate. This results
in strontium sulfide (SrS). SrS seems to be much brighter than CaS. It glows a
greenish colour under UV and has a fairly strong after-glow.
I also tried using strontium carbonate instead of calcium carbonate. This results
in strontium sulfide (SrS). SrS seems to be much brighter than CaS. It glows a
greenish colour under UV and has a fairly strong after-glow.
The picture on the left shows the SrS in the ceramic dish after heating.
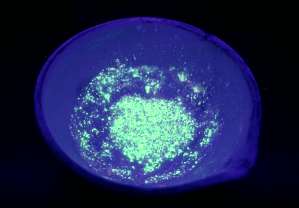 This is the SrS illuminated with a soft UV lamp. It fluoresces a pale yellow / green
colour. It is quite bright and can be seen glowing under UV with the room
lights on.
This is the SrS illuminated with a soft UV lamp. It fluoresces a pale yellow / green
colour. It is quite bright and can be seen glowing under UV with the room
lights on.
In complete darkness, the after-glow is visible for about 15 minutes
Strangely, after storing it in a polythene bag for a few months, it
seems to have become inactive.
Modern luminescent compounds such as zinc sulfide (ZnS) include a controlled quantity
of dopant such as copper. This is written as ZnS:Cu. ZnS:Cu fluoresces brilliant green
under UV light. It is also sensitive to X-Rays and electrons.
 To make CaS, I mixed equal quantities of calcium carbonate and sulfur powder in a
test tube. I also mixed in a very tiny amount (less than 1%) of 'Lo Salt' to act as the
dopant. Lo Salt is manufactured by Klinge and is 66% potassium chloride and 33% sodium
chloride.
To make CaS, I mixed equal quantities of calcium carbonate and sulfur powder in a
test tube. I also mixed in a very tiny amount (less than 1%) of 'Lo Salt' to act as the
dopant. Lo Salt is manufactured by Klinge and is 66% potassium chloride and 33% sodium
chloride.
 I heated this mixture gently over a gas burner until vapor stopped coming off and I was
left with a grey lump in the bottom of the test tube. This lump shows weak luminescence
under UV. I then transferred it to a ceramic dish, ground and mixed it again and then
heated it until it glowed orange. When cool, the compound glows with a mixture of pale
pink and green patches under UV and has an after-glow.
I heated this mixture gently over a gas burner until vapor stopped coming off and I was
left with a grey lump in the bottom of the test tube. This lump shows weak luminescence
under UV. I then transferred it to a ceramic dish, ground and mixed it again and then
heated it until it glowed orange. When cool, the compound glows with a mixture of pale
pink and green patches under UV and has an after-glow. I also tried using strontium carbonate instead of calcium carbonate. This results
in strontium sulfide (SrS). SrS seems to be much brighter than CaS. It glows a
greenish colour under UV and has a fairly strong after-glow.
I also tried using strontium carbonate instead of calcium carbonate. This results
in strontium sulfide (SrS). SrS seems to be much brighter than CaS. It glows a
greenish colour under UV and has a fairly strong after-glow. This is the SrS illuminated with a soft UV lamp. It fluoresces a pale yellow / green
colour. It is quite bright and can be seen glowing under UV with the room
lights on.
This is the SrS illuminated with a soft UV lamp. It fluoresces a pale yellow / green
colour. It is quite bright and can be seen glowing under UV with the room
lights on.